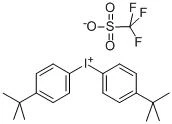
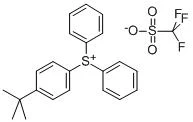
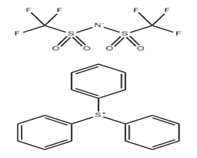
(4-tert-Butylphenyl)diphenylsulfonium triflate (CAS No. 145612-66-4) is a high-performance ionic onium salt primarily utilized as a Photoacid Generator (PAG) in the semiconductor and microelectronics industries (AIP Publishing, 2017). It belongs to the class of triarylsulfonium salts, which are recognized for their exceptional thermal stability and high quantum yield in acid generation upon exposure to specific light wavelengths .
Chemical Properties and Characteristics
The compound consists of a triarylsulfonium cation—specifically one where a tert-butyl group is substituted on one of the phenyl rings—and a triflate (trifluoromethanesulfonate) anion.
- Molecular Formula: C23H23F3O3S2
- Appearance: White to off-white crystalline powder.
- Solubility: High solubility in common organic solvents used in the electronics industry, such as Propylene Glycol Methyl Ether Acetate (PGMEA) and ethyl lactate.
- Thermal Stability: Excellent; triarylsulfonium salts are known for their high decomposition temperatures, which allows them to withstand the "post-apply bake" (PAB) processes in semiconductor manufacturing.
- Photochemical Sensitivity: Highly sensitive to Deep-UV (DUV, 248 nm) and Extreme Ultraviolet (EUV, 13.5 nm) radiation.
Industrial Applications
The primary role of (4-tert-Butylphenyl)diphenylsulfonium triflate is as a critical component in Electronic Chemicals, specifically within the following domains:
- Semiconductor Photolithography
This compound is a staple in the formulation of Chemically Amplified Resists (CARs). When the photoresist is exposed to UV light, the sulfonium salt undergoes photolysis to release a strong Brönsted acid (triflic acid).
- Catalytic Deprotection: The generated acid acts as a catalyst to deprotect the polymer matrix (e.g., removing tert-butoxycarbonyl or tert-butyl ester groups), changing the solubility of the resist in developer solutions.
- Next-Generation Lithography: It is extensively researched for use in EUV lithography, where the efficiency of acid generation directly impacts the sensitivity and resolution of sub-10 nm semiconductor nodes.
- Cationic Polymerization
Beyond semiconductor resists, it serves as a photoinitiator for the cationic polymerization of monomers such as epoxides and vinyl ethers.
- UV Coatings and Inks: Used in high-speed curing processes for industrial coatings where rapid hardening is required under UV irradiation.
- 3D Printing: Utilized in stereolithography (SLA) to initiate the cross-linking of resins.
Mechanism of Action
The efficiency of (4-tert-Butylphenyl)diphenylsulfonium triflate is governed by its photochemical mechanism:
- Excitation: The molecule absorbs a photon, reaching an excited singlet state.
- Photolysis: The sulfur-carbon bond undergoes homolytic or heterolytic cleavage, producing an aryl radical and a radical cation.
- Acid Formation: These intermediates react with hydrogen donors in the resist environment to form Trifluoromethanesulfonic acid (CF3SO3H), one of the strongest known organic acids.
- Amplification: A single photon can trigger a cascade of chemical reactions via this generated acid, significantly increasing the sensitivity of the photoresist system.
Safety and Handling
As an electronic-grade chemical, it must be handled with precision:
- Storage: Should be stored in a cool, dry place, protected from light (to prevent premature acid generation) and moisture.
- Hazards: Typically classified as irritating to eyes and skin. Due to the presence of the triflate group, it should be treated as a precursor to a strong acid.
Product:145612-66-4, Semiconductor Photoresist, Electronic Chemicals

![1138028-44-0,(Z)-2-((Z)-2-((((((1R,4R)-7,7-dimethyl-2-oxobicyclo[2.2.1]heptan-1-yl)methyl)sulfonyl)oxy)imino)thiophen-3(2H)-ylidene)-2-(o-tolyl)acetonitrile](https://www.pudeepharm.com/wp-content/uploads/2025/12/400.jpg)
![1202575-36-7,3-Ethoxy-3-[4-(2-methyl-benzyloxy)-phenyl]-propionic acid](https://www.pudeepharm.com/wp-content/uploads/2025/12/3677.jpg)