Product Description
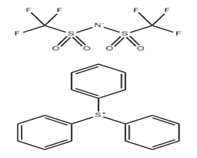
Triphenylsulfonium bis(trifluoromethylsulfonyl)imide, commonly known as TPS-TFSI, is a high-performance chemical compound primarily utilized in the semiconductor and electronics industries as a Photoacid Generator (PAG). Its unique chemical structure allows for high thermal stability and sensitivity to specific wavelengths of light, making it a critical component in the formulation of modern photoresists.
Physical and Chemical Properties
The compound is an ionic salt consisting of a triphenylsulfonium cation and a bis(trifluoromethylsulfonyl)imide anion (TFSI).
| Property | Value/Description |
| CAS Number | 460731-17-3 |
| Molecular Formula | C20H15F6NO4S3 |
| Physical State | Typically a white to off-white crystalline powder or solid. |
| Thermal Stability | High; sulfonium salts generally exhibit superior thermal stability compared to iodonium equivalents, often exceeding 150°C |
| Solubility | Soluble in common organic solvents used in the semiconductor industry, such as Propylene Glycol Methyl Ether Acetate (PGMEA) and Cyclohexanone. |
Industrial Applications
Semiconductor Lithography
TPS-TFSI is extensively used in Chemically Amplified Resists (CARs). In this process, the compound acts as a photo-initiator that decomposes upon exposure to ultraviolet (UV) or extreme ultraviolet (EUV) light to release a strong "superacid".
- Mechanism: The generated acid catalyzes a deprotection or cross-linking reaction within the polymer matrix of the photoresist, which alters its solubility in developer solutions.
- Precision: Its large molecular size and the nature of the TFSI anion help control acid diffusion, which is critical for maintaining high resolution and minimizing "Line Edge Roughness" (LER) in sub-micron circuit patterns.
Electronic Chemicals
Beyond lithography, the compound is categorized as an electronic-grade functional chemical due to its role in:
- Cationic Polymerization: It serves as an initiator for the curing of epoxies and other cationically curable resins used in electronic packaging and 3D printing.
- Antistatic Agents: The TFSI anion is known for providing excellent ionic conductivity and stability, leading to its occasional use in specialized antistatic coatings for sensitive electronic components.
Advantages in the Photoresist Industry
- Non-PFOS/PFOA Compliance: As environmental regulations tighten, the TFSI-based PAGs are often preferred over older perfluorooctane sulfonate (PFOS) based chemicals due to their different environmental profile.
- Sensitivity: TPS-TFSI is highly responsive to deep-UV (DUV) light sources, such as 248 nm (KrF) and 193 nm (ArF) lasers, which are the industry standards for high-volume semiconductor manufacturing.
- Low Volatility: The stability of the TFSI anion ensures that the acid generated does not easily evaporate during the "Post-Exposure Bake" (PEB) process, ensuring consistent pattern transfer.
Safety and Handling
As a specialized electronic chemical, Triphenylsulfonium bis(trifluoromethylsulfonyl)imide should be handled with professional care:
- Storage: Store in a cool, dry place, protected from light to prevent premature decomposition.
- Protection: Use standard laboratory PPE, including nitrile gloves and safety goggles, to avoid skin contact or inhalation of dust.
- Disposal: Must be disposed of as hazardous chemical waste in accordance with local environmental regulations regarding fluorinated organic compounds.

![1138028-44-0,(Z)-2-((Z)-2-((((((1R,4R)-7,7-dimethyl-2-oxobicyclo[2.2.1]heptan-1-yl)methyl)sulfonyl)oxy)imino)thiophen-3(2H)-ylidene)-2-(o-tolyl)acetonitrile](https://www.pudeepharm.com/wp-content/uploads/2025/12/400.jpg)
![1202575-36-7,3-Ethoxy-3-[4-(2-methyl-benzyloxy)-phenyl]-propionic acid](https://www.pudeepharm.com/wp-content/uploads/2025/12/3677.jpg)