Compound Overview and Classification
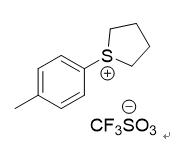
The compound, commonly referred to in industrial settings as p-Tolyltetrahydrothiophenium triflate, is an ionic sulfonium salt. It consists of a cyclic thiophenium cation substituted with a 4-methylphenyl (p-tolyl) group, paired with a trifluoromethanesulfonate (triflate) anion.
In the classification of electronic chemicals, this compound belongs to a critical category known as Photoacid Generators (PAGs)
Chemical and Physical Properties
As a member of the sulfonium salt family, the compound possesses several distinct technical characteristics:
- High Thermal Stability: Sulfonium salts are recognized for their superior thermal stability compared to other initiators, such as iodonium salts, which facilitates their safe storage and processing in industrial environments.
- Latent Reactivity: The compound is chemically "latent," meaning it remains stable and non-reactive under normal conditions (room temperature, absence of specific light wavelengths) but triggers a precise chemical reaction upon irradiation.
- Anion Influence: The use of the triflate anion provides a balance of high acid strength (superacid generation) and controlled mobility within a polymer matrix.
Mechanism of Action: Photoacid Generation
The primary function of this compound in industrial applications is the generation of a strong Brönsted acid upon exposure to specific light sources.
The Photolysis Process
When irradiated—typically by UV or Deep UV (DUV) light—the bond between the sulfur atom and the substituents undergoes homolytic or heterolytic cleavage. This reaction releases trifluoromethanesulfonic acid, one of the strongest known organic acids.
Industrial Applications
- Semiconductor Manufacturing (Photolithography)
This compound is an "indispensable material" in the formulation of Chemically Amplified Resists (CARs) used to manufacture logic and memory chips.
- Sensitivity Enhancement: In a CAR, the acid generated by a small amount of this compound acts as a catalyst, triggering a cascade of deprotection reactions in the resist polymer. This "amplification" allows for high sensitivity, reducing the required light dose and increasing production throughput.
- Resolution and Precision: By modulating the cation structure (the p-tolyl and tetrahydrothiophene components), manufacturers can tune the absorption and diffusion of the acid, enabling the creation of sub-30 nm features required for modern integrated circuits.
- Electronic Chemicals & 3D Printing
Beyond microchips, the compound is utilized in:
- Micro-Electro-Mechanical Systems (MEMS): For high-aspect-ratio patterning.
- Cationic Polymerization: Acting as a photoinitiator for the rapid curing of epoxy resins and coatings used in advanced electronic packaging.
Industrial Standardization and Safety
In the electronic chemicals industry, CAS 2361264-45-9 is produced under stringent high-purity standards. Even trace metallic or ionic impurities can lead to device failure in semiconductor fabrication. Consequently, it is typically supplied in "Electronic Grade" or "VLSI Grade" purity.
Product:2361264-45-9, Semiconductor Photoresist, Electronic Chemicals

![1138028-44-0,(Z)-2-((Z)-2-((((((1R,4R)-7,7-dimethyl-2-oxobicyclo[2.2.1]heptan-1-yl)methyl)sulfonyl)oxy)imino)thiophen-3(2H)-ylidene)-2-(o-tolyl)acetonitrile](https://www.pudeepharm.com/wp-content/uploads/2025/12/400.jpg)