Product Description
"
Alectinib is an oral drug that blocks the activity of anaplastic lymphoma kinase (ALK) and is used to treat non-small-cell lung cancer (NSCLC). Alectinib was approved in Japan in July 2014 for the treatment of ALK fusion-gene positive, unresectable, advanced or recurrent non-small-cell lung cancer (NSCLC).
It was approved by the US Food and Drug Administration (FDA) in December 2015 to treat patients with advanced ALK-positive NSCLC whose disease worsened after, or who could not tolerate, treatment with crizotinib (Xalkori)
It got a conditional approval by the European Medicines Agency in February 2017 for the same indication. This means that additional studies are awaited to confirm a positive benefit-risk-ratio.
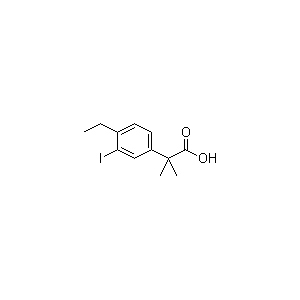
The material 4-Ethyl-3-iodo-alpha,alpha-dimethylbenzeneacetic acid
(CAS Registry Number 1256584-73-2 ) is the key intermediate used for Alectinib.
"

![1138028-44-0,(Z)-2-((Z)-2-((((((1R,4R)-7,7-dimethyl-2-oxobicyclo[2.2.1]heptan-1-yl)methyl)sulfonyl)oxy)imino)thiophen-3(2H)-ylidene)-2-(o-tolyl)acetonitrile](https://www.pudeepharm.com/wp-content/uploads/2025/12/400.jpg)
![2376639-48-2,Ethanesulfonicacid,1,1,2,2-tetrafluoro-2-(1,1,2,2-tetrafluoro-4-tricyclo[3.3.1.13,7]dec-1-ylbutoxy)-](https://www.pudeepharm.com/wp-content/uploads/2025/12/482.jpg)
![1383380-44-6,[1]Benzothieno[3,2-b][1]benzothiophene, 3,8-dibromo-](https://www.pudeepharm.com/wp-content/uploads/2025/12/446.jpg)
![1202575-36-7,3-Ethoxy-3-[4-(2-methyl-benzyloxy)-phenyl]-propionic acid](https://www.pudeepharm.com/wp-content/uploads/2025/12/3677.jpg)