Product Description
"
Abemaciclib is a drug for the treatment of advanced or metastatic breast cancers. It was developed by Eli Lilly. and it acts as a CDK inhibitor selective for CDK4 and CDK6.[1] It was designated as a breakthrough therapy by the U.S. Food and Drug Administration in October 2015.[2]In September 2017, it was approved for use in the United States by the Food and Drug Administration for "adult patients who have hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced or metastatic breast cancer that has progressed after taking therapy that alters a patient’s hormones"
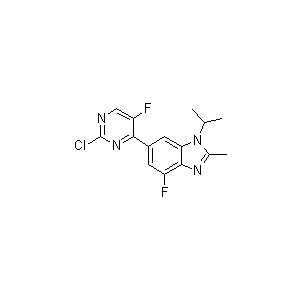
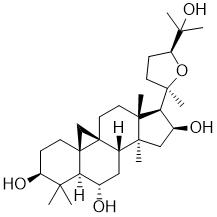
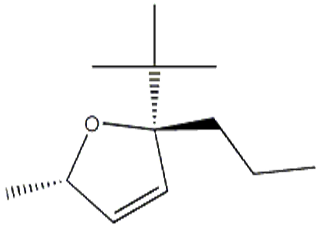
One of intermediates of Abemaciclib, the appearance is light-yellow solid, the purity is not less than 98% by HPLC.
"

![1138028-44-0,(Z)-2-((Z)-2-((((((1R,4R)-7,7-dimethyl-2-oxobicyclo[2.2.1]heptan-1-yl)methyl)sulfonyl)oxy)imino)thiophen-3(2H)-ylidene)-2-(o-tolyl)acetonitrile](https://www.pudeepharm.com/wp-content/uploads/2025/12/400.jpg)
![2376639-48-2,Ethanesulfonicacid,1,1,2,2-tetrafluoro-2-(1,1,2,2-tetrafluoro-4-tricyclo[3.3.1.13,7]dec-1-ylbutoxy)-](https://www.pudeepharm.com/wp-content/uploads/2025/12/482.jpg)
![1383380-44-6,[1]Benzothieno[3,2-b][1]benzothiophene, 3,8-dibromo-](https://www.pudeepharm.com/wp-content/uploads/2025/12/446.jpg)
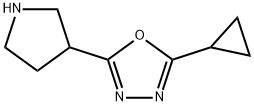
![1202575-36-7,3-Ethoxy-3-[4-(2-methyl-benzyloxy)-phenyl]-propionic acid](https://www.pudeepharm.com/wp-content/uploads/2025/12/3677.jpg)