Product Description
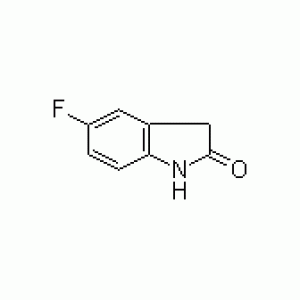
Sunitinib is an oral, small-molecule, multi-targeted receptor tyrosine kinase (RTK) inhibitor that was approved by the FDA for the treatment of renal cell carcinoma (RCC) and imatinib-resistant gastrointestinal stromal tumor (GIST) . Sunitinib was the first cancer drug simultaneously approved for two different indications. 5-fluoroindolin-2-one is a important intermediate to Sunitinib, yellow crystalline powder. Purity 99% +, The minimum order quantity is 1kg. Packing size : 1 kg/bottle, 5kg/bottel, special packing is offered.

![1138028-44-0,(Z)-2-((Z)-2-((((((1R,4R)-7,7-dimethyl-2-oxobicyclo[2.2.1]heptan-1-yl)methyl)sulfonyl)oxy)imino)thiophen-3(2H)-ylidene)-2-(o-tolyl)acetonitrile](https://www.pudeepharm.com/wp-content/uploads/2025/12/400.jpg)
![2376639-48-2,Ethanesulfonicacid,1,1,2,2-tetrafluoro-2-(1,1,2,2-tetrafluoro-4-tricyclo[3.3.1.13,7]dec-1-ylbutoxy)-](https://www.pudeepharm.com/wp-content/uploads/2025/12/482.jpg)
![1383380-44-6,[1]Benzothieno[3,2-b][1]benzothiophene, 3,8-dibromo-](https://www.pudeepharm.com/wp-content/uploads/2025/12/446.jpg)
![1202575-36-7,3-Ethoxy-3-[4-(2-methyl-benzyloxy)-phenyl]-propionic acid](https://www.pudeepharm.com/wp-content/uploads/2025/12/3677.jpg)