Product Description
Enasidenib is an inhibitor of IDH2. It is a drug used to treat relapsed or refractory acute myeloid leukemia in people with specific mutations of the isocitrate dehydrogenase 2 (IDH2) gene. The drug was discovered in 2009 by scientists at Agoios and the development names for the drug was AG-221 and CC-90007.
Enasidenib was approved by the FDA in August 2017. The trade name under which it was approved was Idhifa.
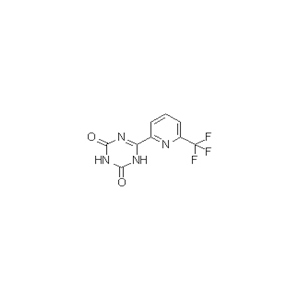
6-[6-(Trifluoromethyl)-2-pyridinyl]-1,3,5-triazine-2,4(1H,3H)-dione is one of KMS of Enasidenib, the appearance is white to off-white powder solid, the purity is not less than 99% by HPLC.

![1138028-44-0,(Z)-2-((Z)-2-((((((1R,4R)-7,7-dimethyl-2-oxobicyclo[2.2.1]heptan-1-yl)methyl)sulfonyl)oxy)imino)thiophen-3(2H)-ylidene)-2-(o-tolyl)acetonitrile](https://www.pudeepharm.com/wp-content/uploads/2025/12/400.jpg)
![2376639-48-2,Ethanesulfonicacid,1,1,2,2-tetrafluoro-2-(1,1,2,2-tetrafluoro-4-tricyclo[3.3.1.13,7]dec-1-ylbutoxy)-](https://www.pudeepharm.com/wp-content/uploads/2025/12/482.jpg)
![1383380-44-6,[1]Benzothieno[3,2-b][1]benzothiophene, 3,8-dibromo-](https://www.pudeepharm.com/wp-content/uploads/2025/12/446.jpg)
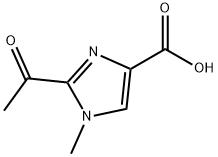
![1202575-36-7,3-Ethoxy-3-[4-(2-methyl-benzyloxy)-phenyl]-propionic acid](https://www.pudeepharm.com/wp-content/uploads/2025/12/3677.jpg)