Product Description
Galangin is a naturally occurring bioflavonoid belonging to the flavonol subclass. It is chemically recognized as a polyphenolic compound widely distributed across specific botanical families and apicultural resources. This article provides a standardized, professional overview of the compound’s physicochemical properties, biological activities, and evolving industrial applications.
Chemical Identity & Physicochemical Properties
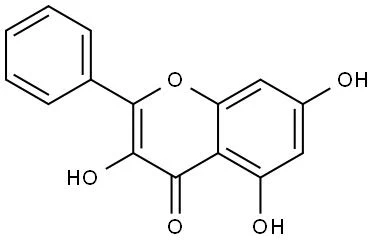
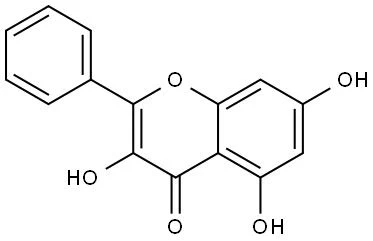
Galangin is structurally classified as a 3,5,7-trihydroxyflavone. Its core scaffold consists of a chromone ring (1-benzopyran-4-one) attached to a phenyl ring, with characteristic hydroxyl substitutions at the C-3, C-5, and C-7 positions.
Physical Appearance:Light yellow crystalline solid or powder
Solubility: Soluble in polar organic solvents (ethanol, DMSO, methanol); sparingly soluble in water.
The arrangement of its m-dihydroxyl structure on the A-ring along with the 3-hydroxy substitution on the C-ring establishes a highly reactive conjugated system capable of intense electronic interaction.
Natural Sources and Biosynthesis
Galangin is synthesized via the secondary metabolic pathways of specific flora. It serves as a primary marker molecule in:
- Rhizomes: Specifically Alpinia officinarum
- Apicultural Products: Propolis and raw honey, where it ranks alongside chrysin and pinocembrin as one of the most prominent polyphenols
- Flora: Helichrysum aureonitens and Uvaria macrophylla leaves.
Biological and Pharmacological Activities
Due to its unique molecular architecture, galangin exhibits robust pharmacological profiles. These mechanisms are evaluated using structural activity relationship (SAR) modeling and density functional theory
- Antioxidant & Radical Scavenging
Galangin acts as a moderate-to-strong antioxidant agent by transferring hydrogen atoms or single electrons to stabilize free radicals. It neutralizes reactive oxygen species (ROS) such as hydroperoxide (OOH) and hydroxyl (OH) radicals, protecting cellular macromolecules (DNA, lipids, and structural proteins) from oxidative stress under physiological conditions.
- Antimicrobial Activity
Galangin possesses pronounced in vitro antimicrobial performance. The compound actively degrades bacterial cell membranes and blocks enzymatic pathways, demonstrating notable efficacy against oral pathogens such as Porphyromonas gingivalis.
- Antiproliferative Effects
In oncology studies, galangin suppresses cancer cell proliferation by modulating cell adhesion and forcing cell cycle arrest at the G2/M or G1 phase. It triggers apoptosis by restoring mitochondrial polarization and down-regulating survival or angiogenic signaling cascades in targeted human cancer cell lines.
Industrial Applications
As industrial manufacturing pivots toward green chemistry and bio-based alternatives, Galangin is experiencing commercial integration across diverse sectors.
- Pharmaceuticals and Nutraceuticals
Because of its therapeutic attributes in handling inflammatory disorders (such as rheumatoid arthritis), galangin is actively processed into natural therapeutic intermediates and dietary supplements designed to regulate cellular communication and boost endogenous antioxidant defense enzymes.
- Functional Cosmetics and Skincare
The cosmetic sector incorporates galangin into advanced topical formulations to shield human keratinocytes from Ultraviolet B (UVB) light exposure. It counters UVB-induced photoaging, mitigates intracellular lipid peroxidation, and rescues skin cells from oxidative degradation.
- Food Science and Active Packaging
In food technology, galangin is leveraged as a natural preservative:
- HAA Mitigation: It actively blocks the generation of harmful heterocyclic aromatic amines (HAAs), such as 2-amino-1-methyl-6-phenylimidazole[4,5-b]pyridine (PhIP), during high-temperature thermal processing of meat products.
- Shelf-Life Extension: Incorporated into biodegradable active food coatings or polymeric films, its antimicrobial action slows decay, preventing spoilage and retaining freshness in perishable foods.