Introduction
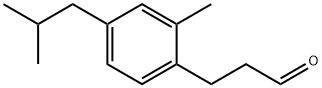
Mahonial is a specialty aroma chemical identified by CAS No. 1536326-17-6. The compound is chemically described as (E)-9-hydroxy-5,9-dimethyldec-4-enal or 4-Decenal, 9-hydroxy-5,9-dimethyl-, (4E)-. Publicly available information on the substance is relatively limited, but existing commercial and chemical database records indicate that it is primarily associated with the fragrance and perfumery industry.
Chemical Identity and Molecular Characteristics
| Property | Information |
| Chemical name | (E)-9-hydroxy-5,9-dimethyldec-4-enal |
| CAS number | 1536326-17-6 |
| Molecular formula | C₁₂H₂₂O₂ |
| Molecular weight | 198.30 g/mol |
| InChIKey | IVCJGAUQMMORNE-YRNVUSSQSA-N |
| Structural features | Unsaturated aldehyde containing a hydroxyl group |
| SMILES | C/C(=C\CCC=O)/CCCC(C)(C)O |
These identifiers are consistently reported across multiple chemical supplier and database references.
The molecule contains both:
- an aldehyde functional group, which is commonly associated with odor-active fragrance molecules, and
- a hydroxyl group, which can influence polarity, reactivity, and olfactory performance.
The presence of an unsaturated carbon chain additionally contributes to its likely fragrance character and formulation behavior. However, detailed peer-reviewed physicochemical studies remain limited in the public domain.
Physical and Chemical Properties
Available published data for Mahonial remain incomplete. The following properties have been reported:
| Property | Value |
| Molecular formula | C₁₂H₂₂O₂ |
| Molecular weight | 198.30 g/mol |
| XLogP3 | 2.2 |
| Hydrogen bond acceptors | 2 |
| Hydrogen bond donors | 1 |
| Exact mass | 198.161979940 |
Publicly accessible sources do not currently provide verified values for:
- boiling point,
- melting point,
- flash point,
- vapor pressure,
- density, or
- refractive index.
Accordingly, such values should not be assumed without validated analytical documentation from manufacturers or regulatory dossiers.
Fragrance and Olfactory Profile
Commercial fragrance suppliers classify Mahonial as an aroma chemical used in perfumery applications. Supplier descriptions associate the material with:
- floral tonalities,
- muguet (lily-of-the-valley) character,
- fresh aldehydic nuances, and
- modern fragrance accords.
The term “Mahonial” itself appears to function primarily as a trade or commercial fragrance name rather than a systematic chemical designation.
Publicly available technical literature does not currently provide a standardized IFRA olfactory monograph or a fully characterized odor threshold profile.
Chemical Reactivity
Based on its molecular structure, Mahonial may participate in several typical organic reactions associated with aldehydes and alcohols:
Oxidation
The aldehyde group may be oxidized to the corresponding carboxylic acid under suitable oxidizing conditions.
Reduction
Reduction reactions may convert the aldehyde moiety into a primary alcohol.
Esterification and Derivatization
The hydroxyl functionality can potentially undergo esterification or etherification reactions useful in fragrance intermediate chemistry.
These reaction pathways are inferred from established organic chemistry principles and are consistent with descriptions in commercial chemical databases.
Industrial Applications
- Fine Fragrance Manufacturing
The principal reported industrial application of Mahonial is in the fragrance sector. Supplier information categorizes the compound under:
- perfume ingredients,
- aroma chemicals,
- scent materials, and
- fragrance intermediates.
Its likely role is as a specialty component for constructing floral and fresh accords in:
- fine fragrances,
- personal care products,
- household fragrance systems, and
- air care formulations.
- Personal Care and Household Products
Commercial references indicate relevance to:
- soaps,
- shampoos,
- detergents,
- fabric care products, and
- cosmetic fragrance systems.
However, publicly available documents do not provide concentration guidelines or formulation limits.
- Aroma Chemical Research and Development
Mahonial is also marketed as a research chemical and synthetic intermediate. Chemical vendors identify it as suitable for laboratory and industrial R&D purposes rather than pharmaceutical or therapeutic use.
Potential uses include:
- fragrance molecule development,
- structure–odor relationship studies,
- synthetic route optimization, and
- specialty fine chemical production.
Environmental and Safety Information
An OECD document listing fragrance materials includes Mahonial among tested substances in aquatic toxicity screening data. One reported entry associates the compound with a zebrafish (Danio rerio) LC50 value of 46 mg/L in the cited dataset.
Nevertheless, comprehensive publicly available regulatory information remains limited regarding:
- occupational exposure limits,
- chronic toxicity,
- biodegradation,
- bioaccumulation,
- reproductive toxicity, or
- long-term environmental behavior.
Users should therefore rely on official Safety Data Sheets (SDS) and supplier regulatory documentation before commercial handling or formulation use.
Conclusion
Mahonial is a specialized aroma chemical identified chemically as (E)-9-hydroxy-5,9-dimethyldec-4-enal. Available evidence indicates that the compound is primarily utilized in fragrance and aroma chemical applications, particularly in floral and muguet-type perfume accords.
Although limited public technical literature exists, currently available sources consistently identify the substance as:
- a fragrance ingredient,
- a fine chemical intermediate,
- a perfumery raw material, and
- a specialty research chemical.
At present, extensive peer-reviewed toxicological and physicochemical datasets do not appear to be publicly available, so technical evaluation should rely on supplier documentation, analytical testing, and applicable regulatory frameworks