Product Description
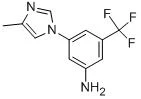
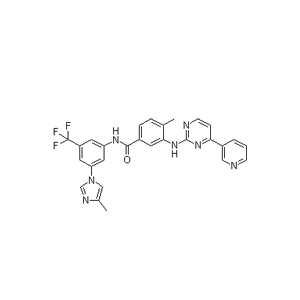
3-(4-Methyl-1H-imidazol-1-yl)-5-(trifluoromethyl)aniline (CAS No. 641571-11-1) is a specialized, highly functionalized aromatic heterocycle primarily utilized as a critical advanced pharmaceutical intermediate. It serves as the core structural precursor in the chemical synthesis of Nilotinib (Tasigna®), a prominent second-generation small-molecule tyrosine kinase inhibitor (TKI) used to treat Philadelphia chromosome-positive chronic myeloid leukemia (CML).
- Chemical Profile and Molecular Properties
The compound’s design strategically positions multiple distinct chemical functional groups on a single benzene core. This creates a highly reactive and specific structural building block necessary for targeting biological systems:
Structural Features
- Trifluoromethyl Group (-CF3): Introduces extreme electron-withdrawing character, enhances metabolic stability, and significantly increases lipophilicity. This adjustment optimizes the final drug's membrane permeability and binding affinity.
- Imidazole Substituent: The 4-methyl-1H-imidazole ring offers crucial hydrogen-bonding capabilities via its nitrogen atoms. This allows the finished active pharmaceutical ingredient (API) to firmly anchor to target protein kinase domains
- Aniline Functionality (-NH2): The primary amine functions as a nucleophilic site, serving as the essential handle for further chemical coupling or chain-extension steps.
- Core Industrial Applications and Uses
The commercial production of CAS No. 641571-11-1 is explicitly driven by the requirements of the pharmaceutical manufacturing sector.
Key Intermediate for Nilotinib Synthesis
The fundamental application of this compound is its use as the final "right-hand" building block in the convergent synthesis of Nilotinib. During industrial production, the primary amine group (-NH2) of 3-(4-methyl-1H-imidazol-1-yl)-5-(trifluoromethyl)aniline undergoes an amidation or coupling reaction with a corresponding substituted benzoic acid or acyl chloride counterpart. This step completes the central amide core of the Nilotinib molecule
Medicinal Chemistry and Derivative Research
Beyond commercial Nilotinib production, the compound serves as an invaluable structural scaffold in drug discovery. Researchers frequently use it to synthesize:
- Novel Schiff Bases: Condensation reactions with aldehydes generate Schiff base derivatives that demonstrate promising in silico binding profiles against alternative oncological targets like Vascular Endothelial Growth Factor Receptor 2
- Next-Generation Kinase Inhibitors: The distinct combination of the imidazole ring and the trifluoromethyl group is a proven pharmacophore model. It provides excellent spatial and electronic geometry for interacting with ATP-binding pockets in mutant or drug-resistant tyrosine kinases
- Industrial Manufacturing and Handling Overview
Industrial Synthesis Pathway
Industrially, this compound is typically synthesized starting from a halogenated 3-trifluoromethyl nitrobenzene or aniline derivative. A nucleophilic aromatic substitution (SNAr) or a transition-metal-catalyzed cross-coupling introduces the 4-methylimidazole ring, followed by the reduction of a nitro group to yield the final primary aniline functionality.
Regulatory and Safety Logistics
As an advanced chemical intermediate, 3-(4-methyl-1H-imidazol-1-yl)-5-(trifluoromethyl)aniline is manufactured under rigorous chemical process controls. It is classified as an industrial chemical irritant requiring standard laboratory and industrial safety compliance, including:
- Local exhaust ventilation to prevent dust inhalation.
- The use of personal protective equipment (PPE) such as nitrile gloves, safety goggles, and protective outerwear.
- Storage in cool, dry conditions under inert gas (such as nitrogen) to protect the primary amine group from atmospheric oxidation over extended periods.