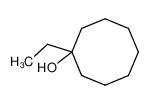
1-Ethylcyclooctanol is an organic chemical compound belonging to the cycloalkanol family. It is characterized by a saturated eight-membered carbon ring (cyclooctane) with an ethyl group and a hydroxyl group both attached to the first carbon atom (C1).
Chemical and Physical Properties
Due to its structure as a tertiary cycloaliphatic alcohol, 1-ethylcyclooctanol typically exhibits the following physical characteristics:
- Molecular Formula: C10H20O
- Molecular Weight: Approximately 156.26 g/mol
- Appearance: Generally a colorless to pale yellow liquid or a low-melting crystalline solid at room temperature.
- Solubility: Exhibits low solubility in water but is highly soluble in organic solvents such as ethanol, diethyl ether, and chloroform.
- Chemical Nature: As a tertiary alcohol, it is resistant to oxidation under standard conditions that would typically convert primary or secondary alcohols into aldehydes or ketones.
Industrial Applications and Uses
The primary industrial utility of 1-ethylcyclooctanol lies in its role as a specialized intermediate within the fine chemical industry, specifically in the following sectors:
- Fragrance and Flavor Synthesis
Cycloaliphatic alcohols are frequently utilized in the fragrance industry due to their unique olfactory profiles. Compounds with the cyclooctyl skeleton often provide:
- Fixative Properties: Helping to slow the evaporation of more volatile scent components.
- Woody or Floral Notes: Serving as a precursor for synthetic musks or woody-scented esters through further esterification processes.
- Chemical Intermediates
1-Ethylcyclooctanol serves as a building block for more complex molecules. In organic synthesis, it can be utilized for:
- Dehydration Reactions: To produce ethylcyclooctenes, which are monomers for specialized polymers or rubbers.
- Esterification: Reacting with various organic acids to create esters used as plasticizers or specialty lubricants.
- Pharmaceutical Research
While not a common active pharmaceutical ingredient (API) itself, it may be used in the synthesis of cycloalkyl-derivative drugs where a rigid, hydrophobic ring structure is required to facilitate specific biological interactions or to alter the lipophilicity of a drug candidate.
Safety and Handling
As with many cycloaliphatic alcohols, standard industrial hygiene practices should be observed:
- Flammability: The compound is combustible. It should be kept away from open flames, sparks, and high-heat environments.
- Storage: Store in a cool, dry, well-ventilated area in tightly sealed containers, preferably under an inert atmosphere (like nitrogen) to prevent slow oxidative degradation.
- Personal Protection: Use chemical-resistant gloves (nitrile or butyl rubber) and safety goggles to prevent skin and eye irritation.
- Disposal: Should be treated as hazardous waste and disposed of according to local environmental regulations, typically through high-temperature incineration.
Product:56470-71-4, cycloalkanol,Cycloaliphatic alcohols