Product Description
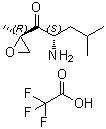
The chemical compound 1-Pentanone, 2-amino-4-methyl-1-[(2R)-2-methyloxiranyl]-, (2S)-, trifluoroacetate is a highly specialized, chiral organic molecule primarily utilized as a vital pharmaceutical building block. It contains a highly reactive and stereospecifically defined alpha-keto-epoxide moiety. This structure serves as the critical "warhead" found in modern second-generation proteasome inhibitors utilized in targeted oncology therapies.
Chemical and Physical Properties
The unique reactivity and application of this compound are governed by its stereochemical configuration and functional groups:
- Molecular Structure: The compound is derived from an L-leucine framework, featuring a 5-carbon ketone chain (1-pentanone) modified with an amino group at the C2 position and a methyl-substituted epoxide (oxirane) ring at the C1 position.
- Stereochemistry: It possesses two crucial chiral centers defined as 2S, 1'R)—or (2S) at the amino-bearing carbon and (2R) at the epoxide ring proximal carbon. Maintaining stereochemical integrity at these positions is vital, as the biological efficacy of the final active pharmaceutical ingredient (API) relies entirely on this specific 3D geometry.
- The alpha-Keto-Epoxide Warhead: The adjacent arrangement of the carbonyl (ketone) group and the epoxide ring creates a highly electrophilic center. This specific arrangement allows it to form irreversible covalent bonds with target proteasomal enzymes.
- Salt Form: It is prepared and isolated as a trifluoroacetate (TFA) salt. The addition of the TFA salt stabilizes the primary amine group, preventing self-condensation or premature ring-opening of the sensitive epoxide ring during storage and handling.
Industrial Applications and Primary Uses
- Core Synthesis of Carfilzomib (Kyprolis)
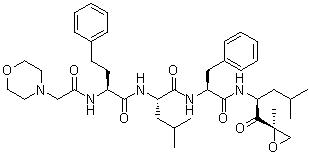
The primary industrial application of CAS No. 247068-85-5 is its use as the key intermediate in the multi-step convergent synthesis of Carfilzomib (trade name Kyprolis). Carfilzomib is a clinically approved, non-boronic acid tetrapeptide epoxyketone proteasome inhibitor used to treat relapsed or refractory multiple myeloma.
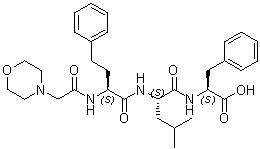
During industrial manufacturing, the free amine of this intermediate is coupled via standard peptide ligation techniques to a tripeptide fragment (Morpholino-carbonyl-Phe-Homophe-Leu-OH) to construct the complete tetrapeptide backbone of Carfilzomib.
- Synthesis of Oprozomib and Next-Generation Proteasome Inhibitors
Beyond Carfilzomib, this structural intermediate is utilized in the development and piloting of other epoxyketone-based proteasome inhibitors, such as Oprozomib (an orally bioavailable developmental analog) .The (2S)-2-amino-4-methyl-1-(2R)-2-methyloxiranyl}]pentan-1-one structural core acts as a broad-spectrum synthone for appending various peptide backbones meant to target the chymotrypsin-like activity of the 20S proteasome core subunit.
Handling, Stability, and Storage
Because the compound contains an active epoxide ring alongside a carbonyl group, it exhibits specific stability liabilities that dictate industrial handling protocols:
- Degradation Pathways: In aqueous or nucleophilic environments, the proximal position of the epoxide ring is highly susceptible to nucleophilic attack, leading to ring-opening degradation into diols or inactive alpha-hydroxy ketones
- Storage Conditions: To prevent over-oxidation, humidity-driven degradation, or epoxide ring-opening, the industrial bulk material must be stored under strict conditions: under an inert gas atmosphere (such as dry nitrogen or argon), at deeply frozen temperatures (typically -20C or below) and completely shielded from moisture.