Product Description
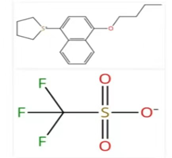
The compound 1-(4-butoxy-1-naphthalenyl)tetrahydrothiophenium 1,1,1-trifluoromethanesulfonate (CAS No. 209482-15-5) is a high-performance Photoacid Generator (PAG). It belongs to the sulfonium salt class of electronic chemicals, specifically designed for use in advanced microlithography within the semiconductor manufacturing industry.
Chemical Structure and Properties
The molecule is composed of a complex organic cation and a stable fluorinated anion:
- Cation: 1-(4-butoxy-1-naphthalenyl)tetrahydrothiophenium. The naphthalene ring provides significant UV absorption characteristics, while the butoxy chain aids in solubility and film-forming properties.
- Anion: Trifluoromethanesulfonate (Triflate). This is a non-nucleophilic anion that ensures the "superacid" generated upon exposure remains highly reactive and mobile.
Key Physical Characteristics:
- Thermal Stability: Designed to withstand the high temperatures used during the Post-Exposure Bake (PEB) process.
- Solubility: Highly soluble in common semiconductor solvents such as PGMEA (Propylene Glycol Methyl Ether Acetate).
- Sensitivity: Optimized for specific wavelengths, particularly deep ultraviolet (DUV) light.
Industrial Applications
This compound is a critical component in the formulation of Chemically Amplified Resists (CARs). Its primary role is in the "Front-End of Line" (FEOL) and "Back-End of Line" (BEOL) stages of semiconductor fabrication.
- Photoresist Industry
In the photoresist matrix, this compound acts as the catalyst. Upon exposure to specific light sources (such as KrF 248nm laser radiation), the sulfonium bond cleaves, releasing trifluoromethanesulfonic acid.
- Semiconductor Lithography
The generated acid migrates through the polymer matrix during the baking process, triggering a cascade of deprotection reactions. This "amplification" allows for:
- High Sensitivity: Lower light doses are required to pattern the wafer.
- High Resolution: Enables the creation of nanometer-scale features necessary for modern integrated circuits (ICs).
- Enhanced Contrast: Provides sharp boundaries between exposed and unexposed regions.
- Electronic Chemicals
Beyond standard lithography, it is utilized in the production of specialized electronic components where precise chemical etching or polymer cross-linking is required.
Safety and Handling
As an ionic photoacid generator, the compound is treated as a sensitive industrial chemical:
- Storage: Must be stored in a cool, dry place, protected from light to prevent premature decomposition.
- Handling: Standard laboratory PPE (gloves, goggles) is required. It is generally handled in "yellow light" environments to prevent accidental activation by ambient UV light.
- Stability: It is stable under normal atmospheric conditions but reacts vigorously with strong bases.

![1242906-47-3,2-Propenoic acid, 2-methyl-, 3,5-bis[(2,2,2-trifluoroacetyl)oxy]tricyclo[3.3.1.13,7]dec-1-yl ester](https://www.pudeepharm.com/wp-content/uploads/2026/05/473.jpg)
![2376639-48-2,Ethanesulfonicacid,1,1,2,2-tetrafluoro-2-(1,1,2,2-tetrafluoro-4-tricyclo[3.3.1.13,7]dec-1-ylbutoxy)-](https://www.pudeepharm.com/wp-content/uploads/2025/12/482.jpg)
![1383380-44-6,[1]Benzothieno[3,2-b][1]benzothiophene, 3,8-dibromo-](https://www.pudeepharm.com/wp-content/uploads/2025/12/446.jpg)
![1202575-36-7,3-Ethoxy-3-[4-(2-methyl-benzyloxy)-phenyl]-propionic acid](https://www.pudeepharm.com/wp-content/uploads/2025/12/3677.jpg)