Product Description
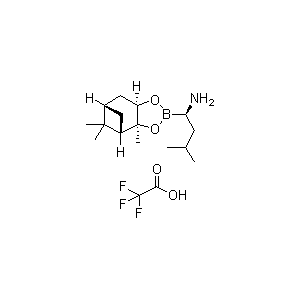
The compound (aR,3aS,4S,6S,7aR)-Hexahydro-3a,8,8-trimethyl-alpha-(2-methylpropyl)-4,6-methano-1,3,2-benzodioxaborole-2-methanamine 2,2,2-trifluoroacetate (commonly referred to as (R)-Boroleucine-(+)-pinanediol ester trifluoroacetate or Bortezomib Amino R-Isomer TFA salt) is a highly specialized organoboron complex. It serves as a cornerstone chiral building block in modern pharmaceutical manufacturing, most notably in the synthesis of oncology therapeutics.
Chemical Structure & Identification
This compound is a stable trifluoroacetic acid (TFA) salt of a chiral amino boronic ester. The structural architecture features a central boron atom protected as a cyclic ester by a (+)-pinanediol chiral auxiliary, adjacent to a stereospecifically defined amino-bearing carbon chain mimicking a leucine residue.
Physicochemical Properties
The compound’s physical behavior is dictated by its dual-nature design: a rigid, lipophilic bicyclic pinanediol core counterbalanced by a highly polar, ionic ammonium trifluoroacetate moiety.
- Appearance: White to off-white crystalline powder or solid.
- Chiral Optical Activity: Highly specific, conforming strictly to the (R)-configuration at the alpha-carbon position relative to the boron center, as dictated by stereospecific induction using the (+)-pinanediol auxiliary.
- Solubility: Exhibiting slight solubility in polar organic solvents like methanol and ethanol. It undergoes specific hydrolytic behaviors depending on pH, which is characteristic of protected boronic esters.
- Stability: Thermally stable as a solid salt form, but sensitive to prolonged atmospheric moisture and oxygen. It requires storage under an inert gas atmosphere (nitrogen or argon) at refrigerated temperatures (2°C to 8°C) to prevent premature deprotection or oxidative degradation of the C–B bond.
Industrial Uses & Pharmaceutical Applications
The compound lacks intrinsic therapeutic application on its own; its absolute value lies entirely in its role as a high-purity pharmaceutical intermediate.
Synthesis of Proteasome Inhibitors
The primary industrial mandate of CAS 179324-87-9 is in the multi-step convergent synthesis of Bortezomib, a first-in-class proteasome inhibitor.
Bortezomib is widely manufactured globally for the treatment of:
- Multiple Myeloma: A malignancy of plasma cells.
- Mantle Cell Lymphoma: An aggressive subtype of B-cell non-Hodgkin lymphoma.
Asymmetric Synthesis and Chiral Reagent Utilities
Beyond Bortezomib, the molecule is utilized within organic development labs as a specialized model or substrate for alpha-aminoboronic acid chemistry. The carbon-boron (C–B) bond acts as a versatile functional handle that can be stereospecifically transformed into various other functional groups (such as alcohols, amines, or homologated alkyl chains) while retaining the chiral integrity installed by the pinanediol group.
Manufacturing Context & Quality Control
In industrial chemistry, this compound is synthesized utilizing Matteson’s asymmetric homologation methodology. Achieving absolute stereochemical purity during this intermediate stage is critical:
- Enantiomeric and Diastereomeric Control: Because therapeutics like Bortezomib require absolute spatial configuration to bind effectively to the 20Sproteasome core, manufacturers tightly monitor this intermediate via chiral HPLC. The presence of the (S)-isomer or epimers at other ring carbon positions must be restricted to minute trace percentages (typically <0.5%).
- Impurity Profiling: Because it is utilized in late-stage active pharmaceutical ingredient (API) synthesis, batches are routinely profiled for trace heavy metals, residual pinanediol, and free trifluoroacetic acid content.
Regulatory, Safety, and Handling Classification
According to globally standardized chemical safety frameworks (GHS), this intermediate must be managed with stringent laboratory controls due to its reactive fluorinated and organoboron components.
- Signal Word: Warning
- GHS Hazard Statements:
- H302: Harmful if swallowed.
- H315: Causes skin irritation.
- H319: Causes serious eye irritation.
- H332: Harmful if inhaled.
- H335: May cause respiratory irritation.
- Handling Protocols: Personnel handling the industrial powder must utilize appropriate Personal Protective Equipment (PPE), including nitrile gloves, chemical splash goggles, and local exhaust ventilation or certified fume hoods. Avoid dust generation and prevent the compound from breaching municipal waterways, as boron-concentrated salts carry elevated aquatic hazard ratings